| Highlights |
Lignin monomer-oligomer separations targeted >90% selectivity. Membranes rejected up to 98.5% of HMW lignin. Up to a 35-fold increase in concentration of LMW products achieved with membranes and RW-EDI. Publications:
|
| Opportunities and Challenges | Lignin is an abundant renewable resource that is a promising substrate for upgrading to biofuels and biochemicals. Depolymerized lignin is a chemically complex stream that consists of aromatic monomers and high-molecular-weight (HMW) oligomers. Regardless of the upstream deconstruction process, separation of monomers and oligomers is a major challenge and opportunity for improved biorefining, given the importance of lignin valorization in lignocellulosic biorefining. Work in the Separations Consortium used an exemplary lignin-rich stream, namely alkaline pretreated liquor (APL), with (1) ceramic membranes to separate the low-molecular-weight (LMW) from the HMW fraction, (2) resin wafer electrodeionization (RW-EDI) to isolate LMW organic acids from APL, and (3) cascading tangential flow filtration (TFF) with ceramic membranes. |
| Membrane Separations | We demonstrated a membrane process to separate APL into HMW and LMW fractions by applying microfiltration (MF) and nanofiltration (NF), or separately, TFF. We characterized the permeance and selectivity of ceramic membranes. The chemical composition, molecular weight distribution, and carbon content of the APL fractions were evaluated to show the impact of filtration. The developed process exhibits up to 98.5% rejection of HMW (MW>1,000 Daltons) species and generates a permeate stream with >80% recovery of LMW lignin-related compounds, including aromatic species such as p-coumarate and ferulate, resulting in a 6-fold enrichment in LMW organic compounds relative to the crude APL (Figure 1). A report of this work was published in Green Chemistry. |
| Techno-Economic Analysis and Modeling of Membrane Processes | A continuous process model was developed to estimate stream flowrates, compositions, and LMW compound recoveries for a two-stage filtration process. Briefly, we used the irreversible thermodynamic model adapted to a multicomponent NF process. The model was developed in Python, and the code is available on GitHub. We used this process model to conduct a TEA to elucidate cost and performance drivers for APL fractionation (Figure 1). Similar TEA was conducted for the cascading TFF study as well. |
| Membrane-Wafer-Assembly Electrodeionization (MWA-EDI) | To overcome the high transfer resistance of commercial ion-conductive membranes for aromatic acids, i.e., p-coumarate and ferulate, a new MWA material was manufactured. We successfully developed the MWA-EDI device and demonstrated an enhancement of aromatic acids capture ratio along with significant reduction of energy consumption and processing cost, as shown in Figure 2. |
| Figures | 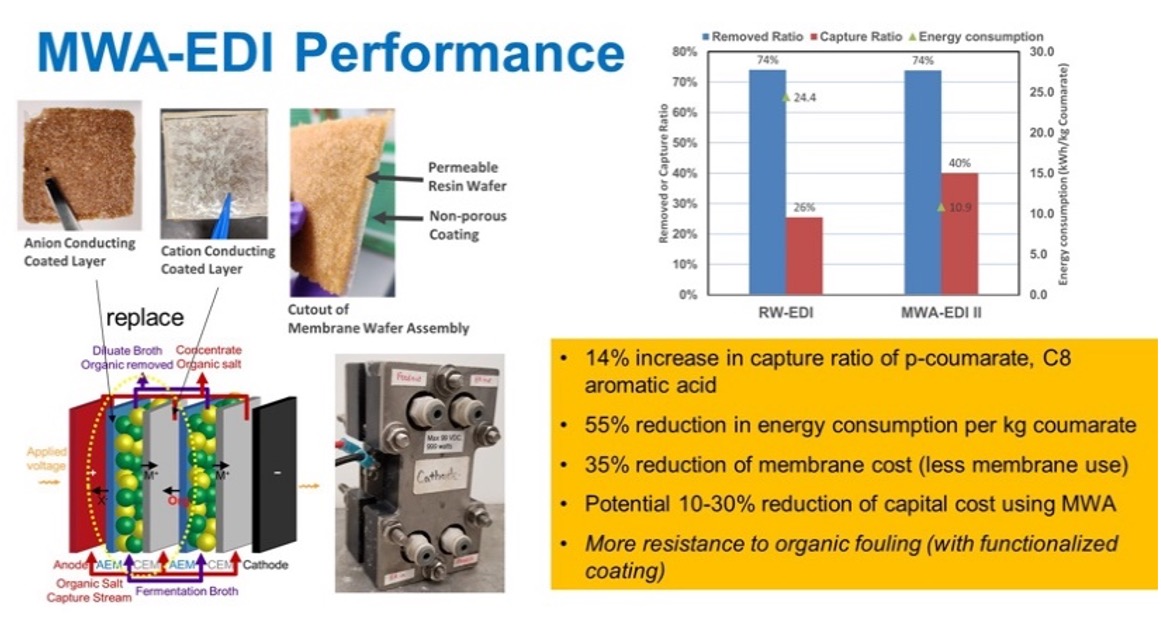 Figure 1. Comparison of separation performance of several membrane options and the resulting LMW distribution in the permeate. 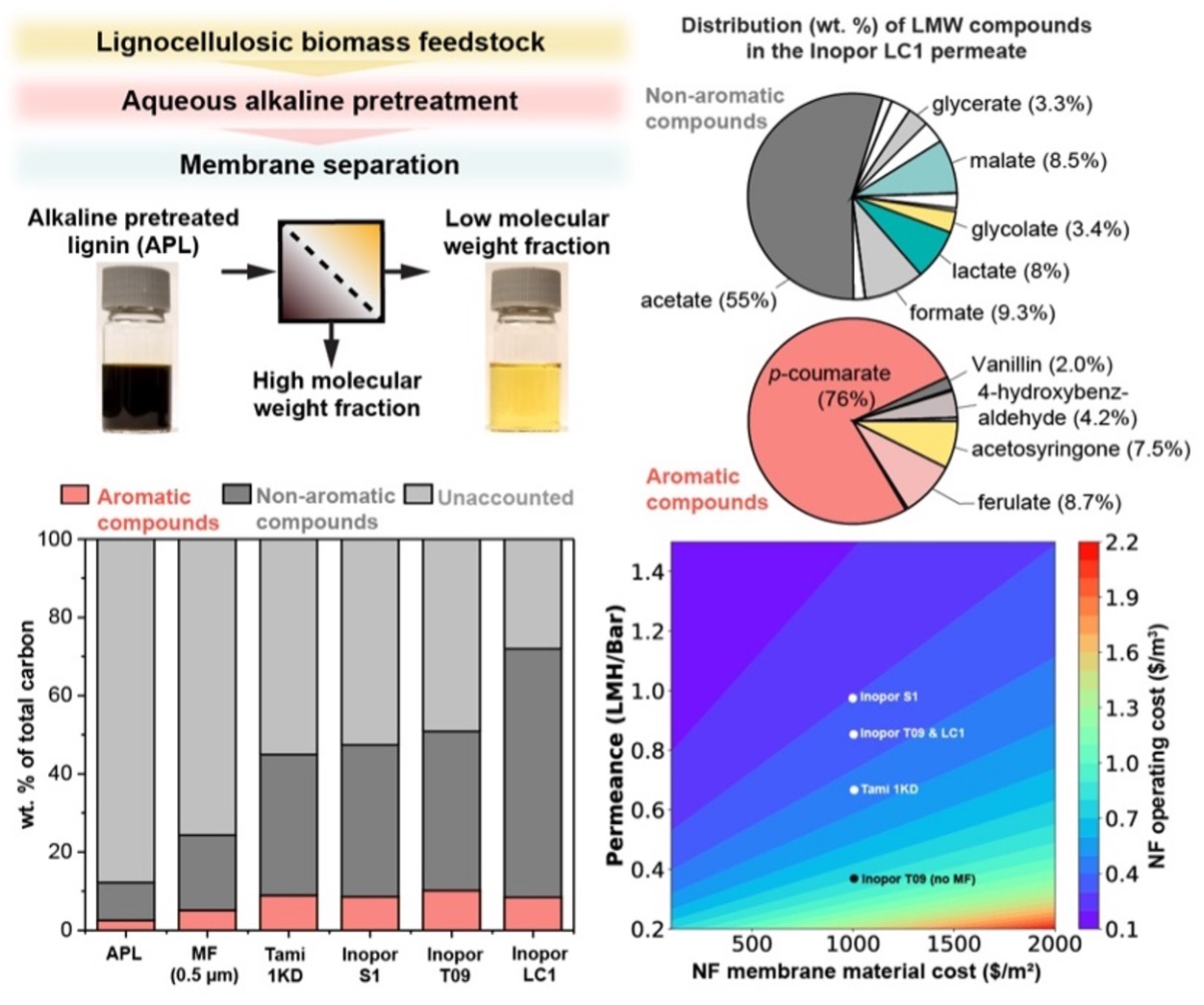 Figure 2. Comparison of separation performance by RW-EDI and MWA-EDI technologies. |
