| Overview |
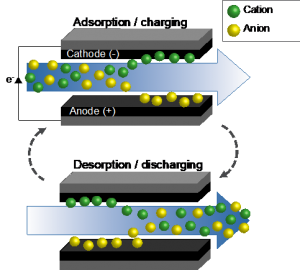
Electrochemical systems for ionic species removal from liquid streams include well-established platforms such as electrodialysis and electrodeionization along with emerging ones such as capacitive deionization (CDI). One advantage of electrochemical separations is the ability to target and remove the minority component (ions) rather than the majority component (water) in a mixture. Based on the principles of electrosorption, CDI offers multiple advantages: 1) lower energy consumption in comparison to separations that require a phase transition, 2) intrinsic reversibility, 3) reagent-free operation, and 4) simple cell design.
In a CDI process, porous conductive electrodes are charged by an applied potential. This causes ions to accumulate in an electrical double layer that forms near the electrode interface. During the charging step, CDI systems are able to store energy, which can then be recovered during the discharge step, reducing the overall cost of the separation processes. Our team is working to advance CDI technology through fundamental material discovery, device-scale fabrication, electrochemical process design and optimization, and process scale-up. Our current focus is on the selective separation of organics (i.e., carboxylates) that are needed for the production of biofuels and bioproducts. This work supports BETO’s mission to develop sustainable bioenergy technologies because energy and cost-effective separations are needed to meet BETO’s biofuel cost targets. Our team is developing the knowledge and tools to enable capacitive deionization for bioprocessing. If successful, our approach for selective separation could be tailored for other applications including homogeneous catalyst recycling, by-product recovery, and impurity removal. |
| National Laboratory | Argonne National Laboratory (ANL) |
| Additional Information | Bioprocesses and Reactive Separations Group at ANL |